
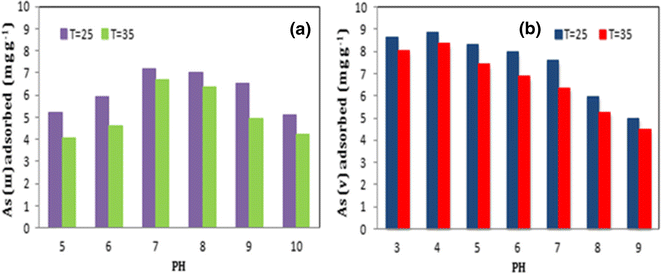
A generic framework, based on the Eigen mechanism, is formulated to describe the formation/dissociation kinetics of inner-sphere metal complexes that may undergo protonation. In principle, all protonated forms of the ligand contribute to the formation of the precursor outer-sphere complexes, but only the sufficiently stable ones effectively contribute to the overall rate of inner-sphere complex formation. The concepts are illustrated by experimental data for Cd(II)-EDTA complexes. Up to pH 8 the dissociation flux in this system is dominated by the protonated inner-sphere complex, even though it is a very minor component of the equilibrium speciation in bulk solution. The results highlight the importance of distinguishing between the thermodynamically predominant species versus the kinetically relevant ones in considerations of dynamic speciation analysis and bioavailability in natural and engineered systems.
To understand the electron transfer mechanisms (outer versus inner sphere) of catalytic superoxide dismutation via a Cu( II / I) redox couple such as occur in the enzyme copper–zinc superoxide dismutase, the Cu( II / I) complexes (L1) 2 Cu(ClO 4) 2 CH 3 CN, ( 1 CH 3 CN) and (L1) 2 Cu(ClO 4 ). What is Inner Sphere Mechanism? Inner sphere mechanism of electron transfer is a redox chemical reaction that occurs via a covalent linkage between oxidant and reductant reactant of the reaction. Here, a ligand bridges the oxidant and reductant reactants during the reaction. However large ligands inhibit this reaction.

